The following article was originally published on September 30, 2014. It has been reviewed and updated as necessary by the ProBrewer editorial staff.
Also see Refrigeration Glossary of Terms and Other Cooling and Heating Equipment in the ProBrewer Classified Ads
By studying and understanding the basic principles of refrigeration, you will be able to understand any type of refrigeration system you might encounter. For any mechanical refrigeration system, the principles and essential components are the same no matter how big or small, or how they are packaged together.
The diagram below illustrates the four basic components of every refrigeration system. These include the Evaporator, the Compressor, the Condenser, and a Metering Device. A refrigerant is then circulated through each component for the sole purpose of removing heat.
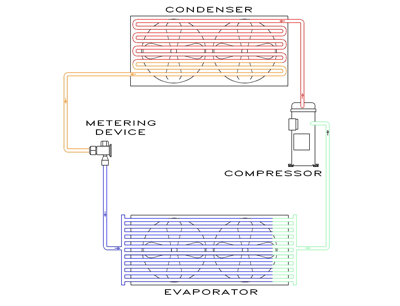
What is Refrigeration? Refrigeration is simply cooling by removing heat. Heat is a form of energy that can’t be destroyed. Therefore to remove heat we can only transfer from one place to another. Even though it is easier to think of refrigeration as the process of making things cold, it actually is the process of transferring heat from one place to another.
Let’s apply this to an example of a walk-in cooler used for the storage of product. In order to cool this room, we need to transfer the heat from inside this room, to somewhere outside this room. This happens in several steps.
Step 1- Air is drawn across a refrigerant Coil, or evaporator, inside the room. As refrigerant flows through this coil, the heat is transferred from the air to the refrigerant. This causes a temperature drop in the air as it passes across the coil.
Step 2- The refrigerant then flows to the condensing unit, where the refrigerant eventually transfers the heat to the outside air at the air cooled condenser. (we will cover the details further on)
Step 3- After the heat is removed from the refrigerant- it then circulates back to the room evaporator to repeat the process.
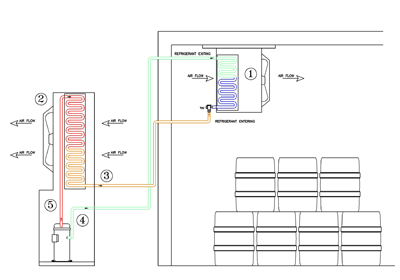
In review, we removed the heat from the room by transferring the heat from the air into the refrigerant (1). The refrigerant carried this heat to the outdoor air cooled condenser, then this heat was transferred from the refrigerant to the outside air (2).
HEAT
To understand the refrigeration process, let’s first make sure we understand Heat.
Heat is a form of energy which can exist on its own and can be moved from one place to another. Heat is not matter that can be measured by weight or volume. Heat can also come from other forms of energy. For example, motors that use electricity will generate heat.
Heat transfer deals with the amount of heat transferred from one place to another. Like all forms of energy, heat flows from a high energy level to a low energy level. One example often used is to think of heat as always traveling “downhill” like water. If water level is the same in two ponds connected by a channel, there will be no water transferred between them. If one pond is higher than the other, the water will flow to the pond at the lower elevation.
Similarly, heat will not flow without a temperature difference. The heat will only transfer from a high energy source (Higher Temperature) to a low Energy Source (Cooler Temperature). The bigger the temperature difference, the more heat is transferred.
There are two kinds of heat, Sensible Heat and Latent Heat.
Sensible Heat is the energy of molecular motion. It is measured by temperature and always causes a temperature change in the substance being heated.
For example, when a pan of water at 32 F is placed on a stove and heated until the temperature climbs to 212 F, it is a sensible heating process. The temperature changed, but the state (liquid) has not. No boiling has occurred.
Latent Heat is the energy of molecular separation and arrangement and cannot be measured with a thermometer. A latent Heat change causes a change of state at a constant temperature.
For example if that pan of water at 212 F is further heated, it will begin to boil. As more heat is added, it will continue to boil until all the water turns to steam (gas) While boiling, the temperature will not increase above 212 F. This is the latent heating process. The temperature doesn’t change, but the state changes from a liquid to a gas.
A characteristic of refrigerant is its ability to boil. That is, to change from a liquid to a gas at a low temperature.
Returning back to our first example of the walk-in cooler. The sensible heat from the room moves into the cool refrigerant as the air is transferred across the cooling coil. This process also removes any latent heat from the air as the water vapor in the air changes state to a liquid and condenses on the cold coil surface (the same way water vapor condenses on the side of a glass of cold beer). The air coming off the coil is not only cooler, but drier than when it entered the coil.
LATENT HEAT – EVAPORATOR
Latent heat transfer is the main way mechanical refrigeration systems move heat. As the refrigerant flows through the coil, and more air comes in contact with the coil, more of the liquid refrigerant boils off until all that remains is a gas.
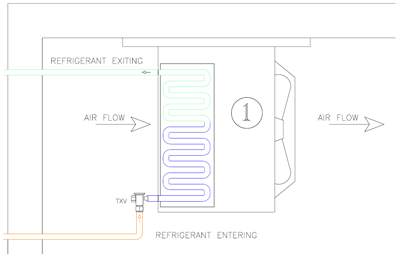
The Latent heat required to boil off this refrigerant from a liquid to a gas is taken from the air as it passes through the coil, thus cooling the air. This boiling process is called evaporation, thus the coil in which this occurs is referred to as our Evaporator. The Evaporator is where we collect the heat we want to remove.
The latent heat process happens at a low temperature because of the nature and properties of the refrigerant and the low pressure in this part of the refrigeration system.
LATENT HEAT – CONDENSER
Refrigerant in the outdoor Air Cooled coil condenses (from a gas to a liquid) at a fairly constant temperature. As the refrigerant flows through the coil, and cooler air is drawn across the coil, more and more of the gas condenses into a liquid until you achieve a solid column of liquid at the outlet of the coil.
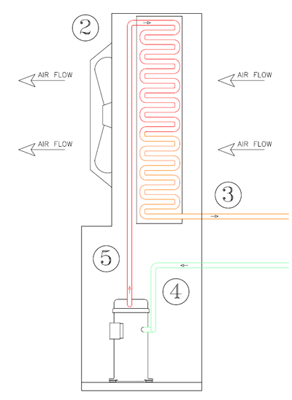
The latent heat required to condense this gas is rejected from the refrigerant into the outside air. This process is called condensation, thus the coil which this occurs is referred to as the condenser. The temperature that the condensation occurs is much higher than within the evaporator, as the pressures in the condenser are much higher.
THE BASIC REFRIGERATION CYCLE
To review, Refrigeration in the simplest terms is cooling by removing heat. It could also be said that Refrigeration is the transfer of heat from one place where it is not wanted (walk in cooler), to another less objectionable place (outdoors).
The normal strategy in mechanical refrigeration is to get the heat to the refrigerant. Then transfer the refrigerant to a place where the heat can be removed from it. The refrigerant makes the heat transfer possible. Presented another way, the refrigerant (R22) is a fluid that picks up heat by evaporating at a low pressure and temperature (within the walk-in evaporator) and then give up this heat by condensing at a higher pressure and temperature (within the outdoor air cooled condenser).

Most of the heat transfer occurs because the refrigerant changes state. The liquid refrigerant in the evaporator absorbs its latent heat of vaporization, and in the process changes from a liquid to a vapor. The gas refrigerant within the condenser rejects its latent heat of vaporization, thus changing from a gas to a liquid. It is this cycle change that moves the removed heat from one place to another.
MECHANICAL REFRIGERATION SYSTEM
We have reviewed the evaporator and the condenser, identifying these two components as the place where the heat is exchanged. Now lets look at the other two key components in the Refrigeration Cycle, the Compressor and the Metering Device.
The compressor and metering device allow two things to happen. The compressor allows us to add mechanical energy to the refrigerant, so that heat can be forced to flow “uphill”. Secondly, they allow us to create two pressure zones in the same system.
In order to keep our product cool, our system needs to absorb heat from within the walk-in cooler. We need to make certain our refrigerant temperature is low enough within the evaporator (evaporation temperature) that the heat we are removing from the walk-in will flow across the coil to be absorbed by the refrigerant.
The normal saturation temperature of the refrigerant within a walk-in cooler evaporator coil is 20 F. Heat will flow down hill from the 40 F air across the coil to the 20 F refrigerant. Since pressure and boiling points are directly related for all fluids, the refrigerant can be made to evaporate at a desired temperature by simply adjusting its pressure to the proper level. For Refrigerant R-22 the saturation temperature at 20 F occurs at 43 PSIG.
Now that we have transferred the heat to the refrigerant, we need to transfer this heat to the outdoor coil. Our system must condense at a high enough temperature that the heat will flow from the refrigerant to the outside air as it passes through the condenser coil. When the outside air temperature is 95 F, the normal saturation temperature for the refrigerant within an air cooled condenser is about 120 F. As in the Evaporator, proper pressure levels will cause the refrigerant to change state at the desired temperature. For R-22 a saturation temperature of 120 F occurs at about 260 PSIG.
To accomplish both the High Pressure and Low Pressure Zones described above, we need to utilize a compressor and a metering device. These two devices mark the dividing points between the high pressure side and low pressure side. Often this is referred to as the High Side and the Low Side of the system.
Please follow the flow of refrigerant as we go through the refrigeration cycle.

The Compressor takes in refrigerant gas at a low pressure and temperature (around 30 F) and compresses it (#4). The discharge off the compressor is on the high side of the system, raising the pressure to about 260 PSIG and about 170 F temperature (#5). The High Pressure/High Temperature gas is then transferred to the air cooled condenser coil (#2). The High Temperature/ High Pressure Refrigerant Gas is then condensed to a high pressure liquid, when the heat is transferred from the refrigerant to the outside air- the liquid temperature has dropped to 120 F, but remains at 260 PSIG. The Condensed Liquid is then transferred from the condenser to the metering device, or TXV (#3). Refrigerant enters the metering device at a high pressure (about 260 PSIG) and exits at a low pressure (43 PSIG) (#1). Here the cycle begins again, with the 20 F liquid refrigerant removing the heat from the 38 F Air within the evaporator. As the refrigerant picks up this heat it boils off from a liquid to a vapor, heating up the refrigerant (to approx. 30 F) while maintaining the 43 PSIG refrigerant pressure. The Heated refrigerant is then transferred to the compressor inlet and recompressed (#4).
In summary, the basic essential components of a mechanical refrigeration system are:
1. An Evaporator to absorb heat into the refrigerant system
2. A Condenser to reject the heat from the refrigerant system.
3. A Compressor to establish the pressures required to force the heat to travel “uphill.”
4. A Liquid Metering Device to regulate the flow of refrigerant and to establish two pressure zones.
This is just a very quick overview of basic refrigeration fundamentals (course material produced by the Carrier Corporation), hopefully it is just enough to help better understand the basic refrigeration cycle. For more in depth information check with equipment providers and take a class.
